Open biomes such as grasslands, savannas, shrublands are associated with many global biodiversity hotspots, and cover ∼60% of land globally. Yet, extensive and increasing anthropogenic activities threaten their functioning and biodiversity. Here, we argue that, in open biomes, researchers and stakeholders (e.g., policy-makers, practitioners) should more comprehensively acknowledge that more than half of a plant’s biomass is typically located belowground. Not only fine roots but different belowground coarse organs of plants (e.g., thick roots, rhizomes) play key ecosystem functions that have been largely neglected in basic and applied ecology. By more accurately accounting for the distribution of these organs along ecological gradients, their biomass turnover and decomposition rate, we would improve estimates of carbon cycling (core in climate change mitigation policies) as well as ameliorating conservation efforts focused on open biomes worldwide.
Grassy and shrubby open biomes – including grasslands, savannas, and shrublands – shaped by recurrent disturbance regimes (e.g., fire, grazing; Durigan and Ratter, 2016), cover ∼60% of land globally (Dinerstein et al., 2017; Ottaviani et al., 2020). Open biomes are also rich in endemic species and thus have a particularly high conservation value (Murphy et al., 2016), and are associated with almost half of the global biodiversity hotspots (Hopper et al., 2021; Myers et al., 2000). Yet, open biomes are experiencing severe threats (Bardgett et al., 2021; Parr et al., 2014; Strömberg and Staver, 2022), which are also linked to the prevailing, and still persisting paradigm that considers them degraded early stages of forest succession, suitable for conversion to intensive agriculture or afforestation (for an overview, see Veldman et al., 2015; Veldman, 2016). The critical importance for ecosystem functioning, climate change mitigation, and biodiversity conservation of open biomes has been historically ignored despite repeated calls by the scientific community (e.g., Bond, 2019; Buisson et al., 2022; Veldman et al., 2015).
Plants in open biomes are adapted to fire, grazing, and/or drought, which can operate as eco-evolutionary forces shaping plant functional strategies (Maurin et al., 2014; Simon et al., 2009). The extent to which these adaptations give plant species in open biomes sufficient capacity to cope with exacerbating environmental conditions and changing regimes – such as more severe fires and heat waves, and rising temperatures – is currently unknown. These adaptations include resource-conservative strategies, characterized by considerable allocation of biomass belowground in specialized coarse organs that can store large pools of carbohydrates (of different types) and shelter buds that can regenerate aboveground biomass after disturbance (e.g., Ottaviani et al., 2020; Pausas et al., 2018; Simon et al., 2009). These plant organs and related strategies promote key ecosystem functions, including biomass production, soil stabilization, and carbon sequestration in the soil (Klimešová et al., 2018, 2021, 2023; Ottaviani et al., 2021; Teixeira et al., 2022). Nevertheless, belowground coarse organs (BCOs) have been largely overlooked in basic and applied ecology as well as in climate change mitigation research.
In this piece, BCOs refer to any plant organ located belowground, other than fine roots, (e.g., thick roots, rhizomes, lignotubers, xylopodia, bulbs; Klimešová et al., 2018). We use BCOs inclusively, because our aim is to call for a broader assessment of the importance of BCOs in open biomes’ dynamics, functioning, and biodiversity conservation, rather than to redefine well-established terms and notions in the literature – such as belowground bud bank and clonal organs (Klimešová et al., 2019; Pausas et al., 2018) or underground storage organs (Wigley et al., 2020).
We address the relevance of open biomes for ecosystem functioning, with a particular reference to the core function of soil carbon cycling and the role played by plant BCOs. We discuss how underestimating the belowground dimension (e.g., by focusing on fine roots only) can undermine our capacity to assess and value ecosystem functioning as well as to support conservation actions in open biomes. Finally, we provide our perspective on the need to gather more realistic and accurate estimates of the contribution of all belowground organs to ecosystem functioning in globally distributed open biomes.
Digging deeper (and coarser) into the soil carbon cycling of open biomesThere is growing recognition that open biomes play major roles in carbon cycling globally (Bengtsson et al., 2019; Zhao et al., 2020). Particular attention has been devoted to belowground carbon storage and sequestration to explore the potential of grasslands, savannas, and shrublands in mitigating climate change. For example, a recent study estimated that grasslands account for nearly a third of global terrestrial carbon stocks (Bai and Cotrufo, 2022). It is now widely acknowledged that carbon storage in open biomes is chiefly happening belowground (Fidelis et al., 2013; Zhou et al., 2022), therefore carbon cycling could only be poorly assessed by remote sensing (Cavender-Bares et al., 2022). For example, grassland soils contain 80–94% of the total carbon pool as soil organic carbon and in plant organs located belowground (Liu et al., 2021).
Nevertheless, studies examining plant-soil interactions and their effects in the carbon cycle are often directed towards fine roots only, overlooking the contribution of BCOs in carbon storage and cycling (see e.g., Bai and Cotrufo, 2022). BCOs perform multiple key functions for the plant, such as (1) storage of carbohydrates and buds for sprouting after seasonal rest and regeneration after major disturbances (e.g., drought, fire, herbivory), (2) space exploration and occupancy, (3) resource absorption by determining the location of fine roots, and (4) anchorage in the soil (Bell and Tomlinson, 1980; Klimešová et al., 2018). BCOs can account for a substantial component of plant community biomass in open biomes (Mokany et al., 2006; see Table 1), which is often higher than that of fine roots (Blume-Werry et al., 2018) and aboveground biomass (Ottaviani et al., 2020; Table 1), and are integral to belowground litter and carbon cycle. Despite their relevance, BCOs are understudied in plant ecology at large (compared to stems, leaves, seeds, or fine roots; Klimešová et al., 2020; Laliberté, 2017), and their role in carbon cycle is rarely examined even though the mechanisms and decomposition rate can differ greatly between belowground plant organs (e.g., Amougou et al., 2011). This constitutes, in our opinion, a significant gap that needs to be better addressed in future studies and policies.
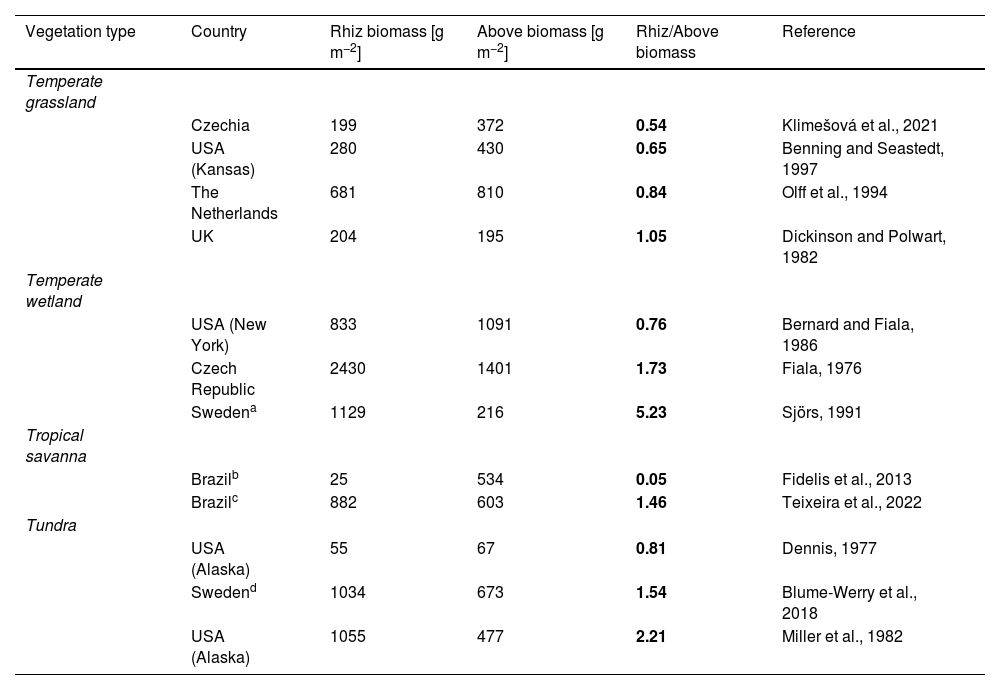
Examples of mean values and ratios of community-level rhizome (Rhiz) and aboveground (Above) biomass data in four vegetation types (in italics) from open biomes worldwide. Vegetation types are ordered alphabetically, and within them each study is sorted by an ascending order of Rhiz/Above biomass ratio (in bold).
| Vegetation type | Country | Rhiz biomass [g m−2] | Above biomass [g m−2] | Rhiz/Above biomass | Reference |
|---|---|---|---|---|---|
| Temperate grassland | |||||
| Czechia | 199 | 372 | 0.54 | Klimešová et al., 2021 | |
| USA (Kansas) | 280 | 430 | 0.65 | Benning and Seastedt, 1997 | |
| The Netherlands | 681 | 810 | 0.84 | Olff et al., 1994 | |
| UK | 204 | 195 | 1.05 | Dickinson and Polwart, 1982 | |
| Temperate wetland | |||||
| USA (New York) | 833 | 1091 | 0.76 | Bernard and Fiala, 1986 | |
| Czech Republic | 2430 | 1401 | 1.73 | Fiala, 1976 | |
| Swedena | 1129 | 216 | 5.23 | Sjörs, 1991 | |
| Tropical savanna | |||||
| Brazilb | 25 | 534 | 0.05 | Fidelis et al., 2013 | |
| Brazilc | 882 | 603 | 1.46 | Teixeira et al., 2022 | |
| Tundra | |||||
| USA (Alaska) | 55 | 67 | 0.81 | Dennis, 1977 | |
| Swedend | 1034 | 673 | 1.54 | Blume-Werry et al., 2018 | |
| USA (Alaska) | 1055 | 477 | 2.21 | Miller et al., 1982 |
This study deals with an open fen, which we consider here to belong to wetlands, in a broader sense.
This study separates roots (including fine and thick ones) vs other belowground organs (e.g., rhizomes, bulbs).
We highlight here three main reasons why BCOs should be taken into account to better understand their contribution and potential effects on the overall carbon cycle in open biomes. We use rhizomes as an example because these organs are very common across species forming grassy and shrubby biomes, and therefore tend to be more studied than tubers, lignotubers, xylopodia, or bulbs (but see Meller et al., 2022; Pausas et al., 2018; Tsakalos et al., 2022). However, the same reasoning applies to the other BCOs. First, rhizomes may account for a conspicuous amount of plant biomass at the community level in open biomes that may equal or exceed aboveground biomass (Table 1). Rhizome biomass of an individual plant increases during establishment until it reaches maturity (Bell and Tomlinson, 1980). Ancient open ecosystems may host old, developed, large individual plants with rhizomes of remarkable biomass that has been accumulated over several growing seasons (Buisson et al., 2022). Rhizome biomass may scale linearly with aboveground biomass (slope of the scaling relationship ∼1; Ottaviani et al., 2021), possibly due to accumulation over seasons being balanced by changes in decomposition rate with age (for herbs, see Harris et al., 2023), and the rhizome:aboveground biomass ratio can be highly species-specific. Second, the perenniality of BCOs may vary across environmental gradients. For example, rhizomes tend to be more persistent with a slower biomass turnover under drier and more nutrient-limited conditions, which may lead to a higher standing rhizome biomass in arid and low-productive temperate grasslands (Klimešová et al., 2018, 2023). Additionally, rhizomes contribute to soil organic carbon fraction and litter decomposability differently than roots because of different tissue composition between these belowground organs (hence recalcitrance to decomposition; Amougou et al., 2011). Third, rhizome biomass can be markedly reduced by even slight increases in grassland management intensity (Ottaviani et al., 2021) – with implications for other plant and ecosystem functions specifically provided by rhizomes, such as storage of carbohydrates and buds for vegetative regeneration or protection against erosion (Klimešová et al., 2023), and for species diversity (Lisner et al., 2021). In tropical savannas, where shrub abundance is higher, the relationship between biomass allocation strategies, management, and ecosystem functioning may differ (Fidelis et al., 2013; Teixeira et al., 2022).
Improving assessments of belowground functioning and conservation actions in open biomesStandardized protocols to identify BCOs and collect data on these organs are becoming increasingly available (e.g., measuring traits; Klimešová et al., 2019; Pausas et al., 2018; Wigley et al., 2020). These approaches can be readily implemented to improve the accuracy of carbon flux estimates, such as using traits to estimate biomass allocation strategies in different plant organs (e.g., Klimešová et al., 2021). Multiple lines of evidence indicate that incorporation of BCOs contributes to a broader understanding of carbon cycle in open biomes. However, accurate estimates of biomass allocated to BCOs are often missing from the literature (e.g., Bai and Cotrufo, 2022), and particularly in tropical grasslands and savannas, where they play key functional roles (Teixeira et al., 2022). The process of providing benchmarks, against which the outcomes of climate-change mitigation or conservation actions can be compared, may benefit from including summaries of the belowground biomass allocation to different organs in healthy ecosystems – considering that relative abundance and biomass of different BCOs and fine roots can change along environmental gradients (Blume-Werry et al., 2018; Klimešová et al., 2023).
ConclusionsEcosystem functions and biodiversity of open biomes have been historically undervalued by scientists, policy-makers, and the general public. Here, we call for greater consideration of the importance of BCOs in playing key, yet overlooked roles to support nature and people in open biomes worldwide. BCOs take a long time to become fully developed, considerably longer than the time needed for establishment of fine roots (which have a quicker biomass turnover than BCOs), stressing the relevance of protecting ancient open biomes (Buisson et al., 2022; Nerlekar and Veldman, 2020). We argue that these differences in the rate of biomass accumulation and decay should be better considered to design more accurate and effective climate mitigation policies and conservation actions. This calls for rethinking the timing at which the ecosystem health and the management practices are monitored and assessed in open biomes. Otherwise, these will likely fail to deliver the expected outcomes for soil carbon stock and sequestration as well as for biodiversity at the local and global scale.
Authors’ contributionGO and FAOS conceived the research idea and led the writing of the manuscript. All coauthors contributed to developing the idea and to revisions.
This work was supported by the Czech Science Foundation (grant number: 19-13231S) and by the long-term research development project of the Czech Academy of Sciences (RVO 67985939). GO acknowledges the support by the NBFC (National Biodiversity Future Center) funded by the Italian Ministry of University and Research, P.N.R.R., Missione 4 Componente 2, “Dalla ricerca all’impresa”, Investimento 1.4, Project CN00000033 (funded by the European Union – NextGenerationEU). JK. MM and TH acknowledge the support by the Ministry of Education, Youth and Sports of the Czech Republic (LTT20003). We thank the Associate Editor (Gerhard Overbeck) and an anonymous Reviewer for providing useful suggestions that improved the clarity and quality of this manuscript.