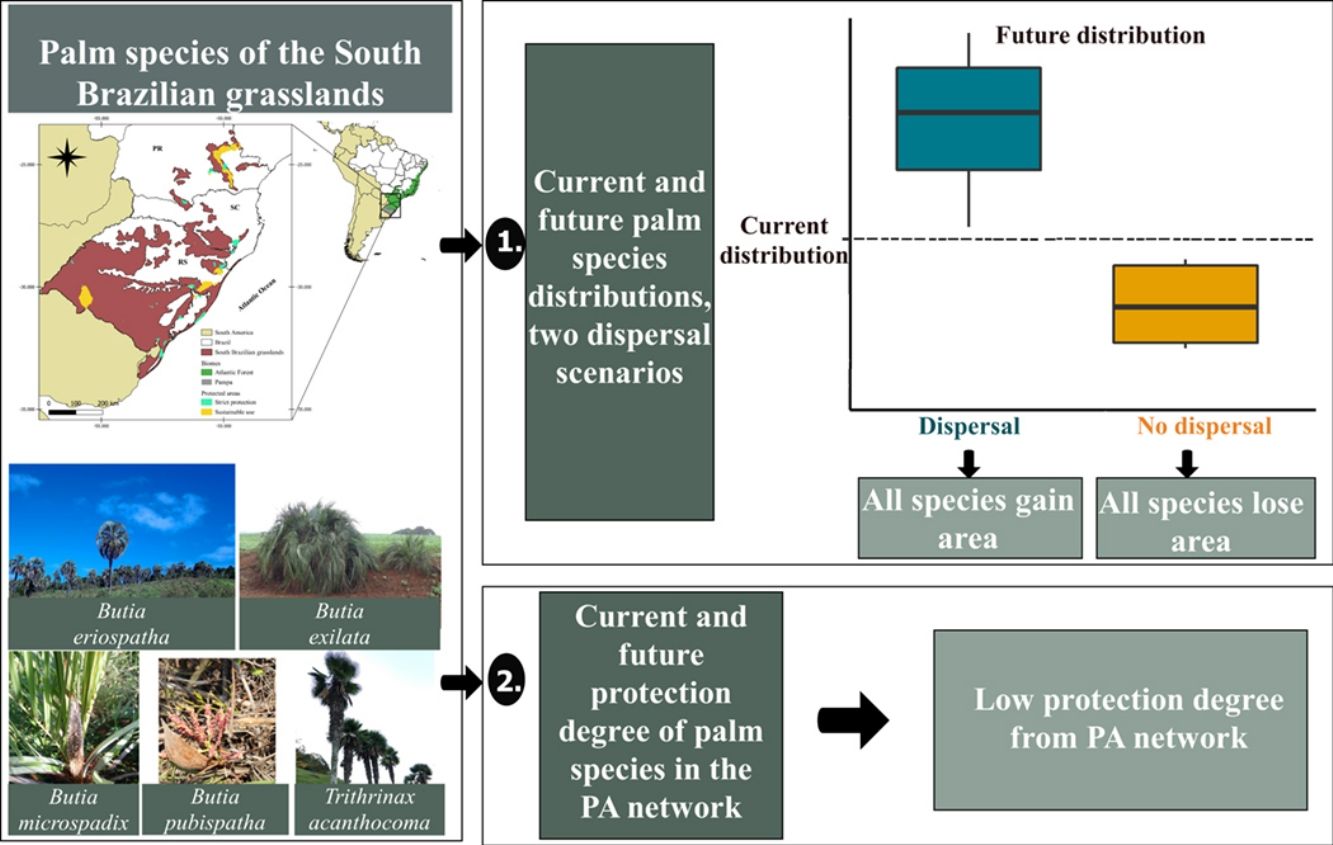
South Brazilian grasslands are at risk due to climate change and activities that convert native vegetation into agricultural lands. We used 12 native palm species as ecological indicators of these ecosystems to evaluate the impact of climate and land-use changes on their geographical distribution. We estimated the potential distribution of species for current and future (year 2050; RCP 4.5 and 8.5) climate and land-use conditions considering two dispersal scenarios. We also assessed the coverage of protected areas (PAs) for current and future species distribution. Our results indicated that the future distribution of all species will decrease under scenarios with no dispersal and increase under scenarios with dispersal; however, a dispersal scenario might be unlikely considering species dispersal ability and the increased pressure on grasslands. A comprehensive strategy is fundamental to ensure species conservation and recovery since the current PAs network covers less than 2% of the current distribution for nine out of the 12 species. Assessing the synergy of trends in climate and land-use changes is essential to improve conservation strategies for these emblematic species.
The interaction of climate and land-use changes leads to synergistic effects that drive the decline of biodiversity and ecosystem services (Brook et al., 2008; He et al., 2019). Synergistic effects are factors that interact and increase the extinction risk of a species (Brook et al., 2008). Because rapid climate change may hamper adaptive responses in many species, sufficient dispersal is often the only mechanism preventing extinction (Garcia et al., 2014; Loarie et al., 2009; Parmesan, 2006). However, species dispersal ability in response to climate change may be compromised by habitat loss and fragmentation, which impose barriers for many species (Brook et al., 2008; He et al., 2019; Sales et al., 2019). Among plants, species with abiotically-dispersed seeds which establish viable populations reaching reproductive maturity in a relatively short time (Cramer et al., 2007; Di Musciano et al., 2020; Tabarelli and Peres, 2002), may be able to persist in the face of current habitat loss and fragmentation. On the other hand, species with large zoochoric fruits and seeds have slower dispersal rates, are less likely to cope with environmental changes and, consequently, are more prone to extinction (Cramer et al., 2007; Tabarelli and Peres, 2002). Therefore, it is important to consider the potential synergies of climate and land-use changes, especially for species with limited dispersal, which can be done by modeling the distribution of species (Lemes and Loyola, 2015) and assessing the effectiveness of protected areas (PAs) in the face of those threats.
A major strategy in conservation has been implementing PAs, which seek to maintain biodiversity and ecosystem functioning through time (Margules and Pressey, 2000). PAs may not protect resident species that change their distribution in response to climate change (McDonald et al., 2019), especially in a country like Brazil, where PAs are unevenly distributed across biomes (Overbeck et al., 2015b; Vieira et al., 2019). A few studies have recently analyzed the impact of future climate change and habitat loss on the distribution of different organisms in an integrative way, including woody plants from the Atlantic Forest (Zwiener et al., 2017), plants from the Cerrado (Velazco et al., 2019), and monkeys from the Amazon (Sales et al., 2019). While these biomes are usually at the core of the conservation agenda, other biomes like Pantanal, Caatinga, and Pampa are usually neglected (Vieira et al., 2019). Specifically, open native ecosystems such as the South Brazilian grasslands are severely threatened, poorly known, and receive insufficient attention in the conservation agenda (Overbeck et al., 2007, 2015b).
South Brazilian grasslands are natural open ecosystems occurring under subtropical climate in the Pampa and Atlantic Forest biomes in Brazil's southern states (Overbeck et al., 2015a). These grasslands are threatened due to conversion for agriculture and silviculture (Overbeck et al., 2007; Vélez-Martin et al., 2015), and by climate change (Costa et al., 2018). Paleopalynological studies showed that the grassland-dominated landscape of southern Brazil has started to be subject to forest expansion promoted by increased temperature and humidity in the late Holocene (Behling, 2002), evidencing that grasslands are relictual ecosystems in the region. The potential for forest expansion over grassland is expected to be maintained along the 21st century (Anadón et al., 2014; Salazar et al., 2007). To understand the effects of climate and land-use changes in the South Brazilian grasslands, we can use typical species of these ecosystems as ecological indicators and project their modeled distribution into future climate change scenarios.
In the South Brazilian grasslands, several palms are part of the relictual vegetation and play a fundamental ecological role as food resources for the local fauna (De Lourdes et al., 2007; Pintaud et al., 2008; Sosinski et al., 2019). Although the distribution of palm species (Arecaceae) is predominantly pantropical, species of genera such as Allagoptera, Butia, and Trithrinax have established in the grassland ecosystems of South America (Pintaud et al., 2008), where the combination of climatic, edaphic, and historical factors promoted their diversification (Bjorholm et al., 2006a,b). Palm species of these genera are distributed throughout the South Brazilian grasslands (Soares et al., 2014) and these palms may be considered as ecological indicators for these ecosystems. Understanding how these palms could respond to future climate and land-use changes may help us predict the future of the South Brazilian grasslands and provide informed conservation strategies. We addressed the following questions: (1) what are the impacts of climate and land-use changes on the geographical distribution of palm species in the South Brazilian grasslands given two dispersal scenarios: without dispersal (hereafter, “no-dispersal”) or with dispersal (hereafter, “dispersal”)?, and (2) to what extent are these species encompassed by the current network of protected areas in present and future distribution? We hypothesized that (1) in the dispersal scenario the distribution area of palm species will increase because the synergistic effects of climate and land-use changes will not be sufficient to prevent species dispersal to new areas, (2) in the no-dispersal scenario, the distribution area of palm species will decrease due to synergistic effects of climate and land-use changes, because barriers resulting from these changes will prevent species dispersal to new areas, and (3) the studied palm species will be poorly covered by PAs in the present and future due to the small total area of the PA network in South Brazilian grasslands.
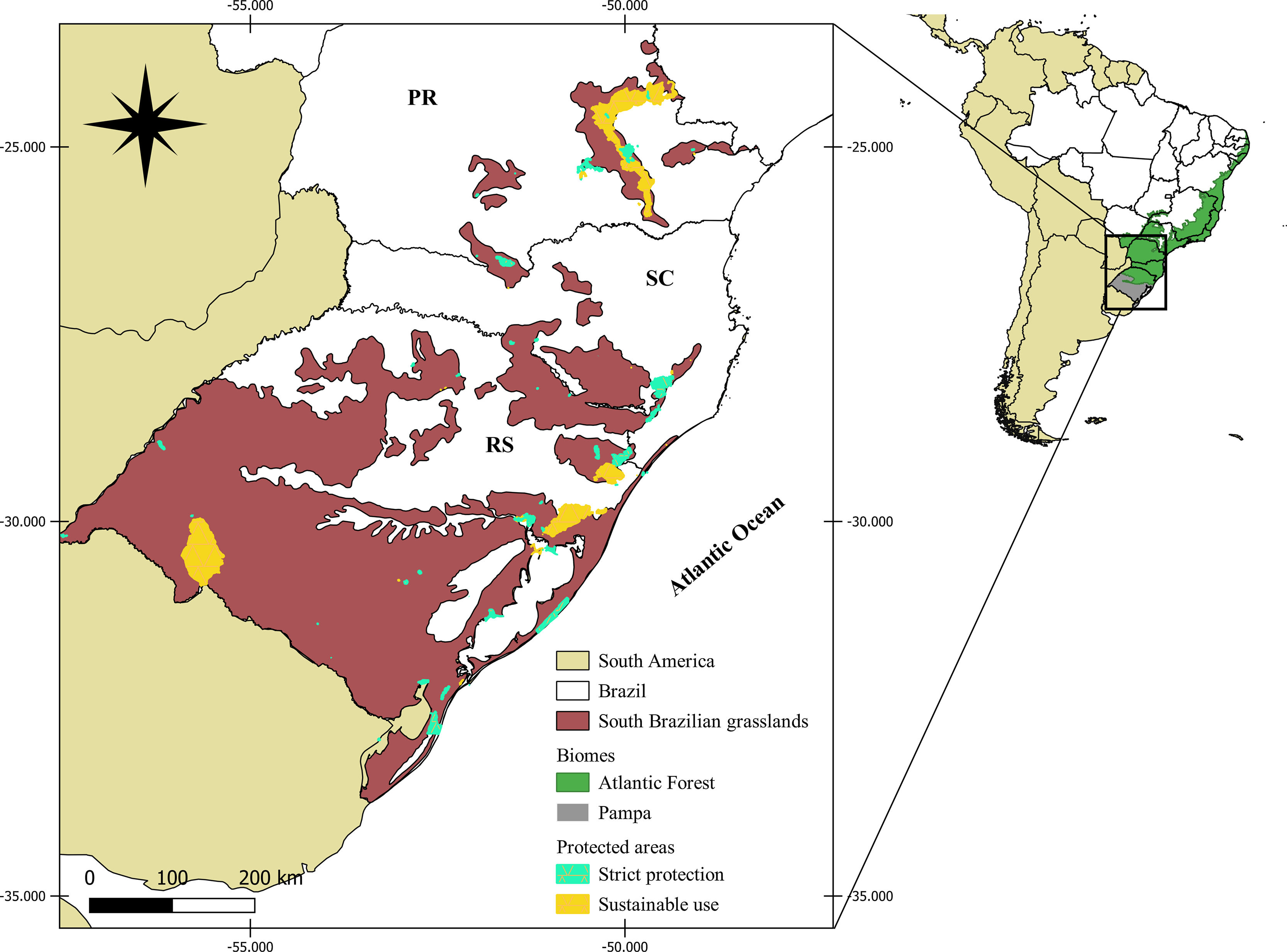
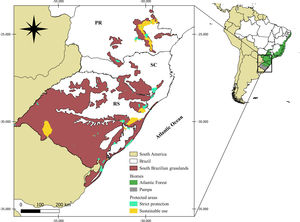
MethodsStudy regionThe South Brazilian grasslands (Fig. 1) encompass the Brazilian Pampa and a portion of the Atlantic Forest biomes (Overbeck et al., 2015a, 2007). Climate is humid subtropical, with mild summers in the Atlantic Forest and hot summers in the Pampa (Alvares et al., 2013). Grazing and fires are disturbances that maintain plant diversity in South Brazilian grasslands, provided that appropriate management techniques are used (Overbeck et al., 2015a).
Map of the South Brazilian grasslands and their protected areas. Protected area data obtained from CNUC (2019). Map adapted from (Overbeck et al., 2007, 2015a).
The list of palm species of South Brazilian grasslands was obtained from the Brazilian Flora platform (http://floradobrasil.jbrj.gov.br/), where we searched for native palm species that occur in Campos de Altitude, Campo Limpo, and Campo Rupestre associated with the Atlantic Forest and Pampa biomes in the states of Paraná (PR), Santa Catarina (SC), and Rio Grande do Sul (RS) (Fig. 1). Hybrids and subspecies were not included. We found 12 palm species, which we classified into those restricted to grassland ecosystems (hereafter “typical palm species”), and those that also occur in other vegetation types and biomes (e.g., Cerrado; hereafter “associated palm species”). The typical palm species are Butia eriospatha (Mart. ex Drude) Becc, Butia exilata Deble & Marchiori, Butia lallemantii Deble & Marchiori, Butia microspadix Burret, Butia odorata (Barb.Rodr.) Noblick, Butia pubispatha Noblick & Lorenzi, Butia witeckii K. Soares & S. Longhi, and Butia yatay (Mart.) Becc. The associated palm species are: Allagoptera campestris (Mart.) Kuntze, Butia paraguayensis (Barb. Rodr.) Bailey, Trithrinax acanthocoma Drude and Trithrinax brasiliensis Mart. (see species conservation status in Table S1).
Species occurrencesWe obtained species occurrences from REFLORA (http://reflora.jbrj.gov.br/), SpeciesLink (http://www.splink.org.br/), GBIF (https://www.gbif.org/), BIEN (http://bien.nceas.ucsb.edu), JABOT (http://rb.jbrj.gov.br/), and new fieldwork records. For species with <20 occurrences, we considered the geographical coordinates from municipality centroids, which have not biased distribution models for plants in the Cerrado (Velazco et al., 2019). We included occurrences of synonymous species, which happened only for B. lallemantii and T. acanthocoma (more details on Supplementary Data, Appendix S1). We filtered occurrences based on a grid with a 2.5min resolution to reduce bias in occurrence sampling by selecting one occurrence per pixel, totaling 579 occurrences, and ten species used for species distribution modeling (Table S2, Figs. S1 and S2). We built distribution models only for species with >5 occurrences, thus B. pubispatha and B. witeckii were disregarded.
Environmental data and climatic scenariosWe used climatic and soil environmental variables, which are important to characterize terrestrial plant species niche (Velazco et al., 2017). Climatic variables were obtained from CHELSA v.1.2 at a resolution of 2.5min (c. 4.5km; http://www.paleoclim.org/), because this dataset contains better estimates of the climatic variables (Karger et al., 2017) and produced models with better performance than Worldclim (Bobrowski and Udo, 2017). Soil physical variables were obtained from Soil Grids database (Hengl et al., 2017) at a resolution of 0.75 arcseconds (c. 250m; ftp://ftp.soilgrids.org/) and upscaled to the same resolution as climatic variables. Species distribution models were constructed at this resolution due to computational limitations. Climatic and soil variables comprised 56 environmental layers (Table S3). We obtained future climate projections for the year 2050 from seven global circulation models (AOGCMs) which account for the highest variability for the Neotropical region (Velazco et al., 2019): CESM1-BGC, CSIRO-ACCESS-1.3, FIO-ESM, GFDL-ESM2G, GISSE2-R, IPSL-CM5A-LR, and MOHC-HADGEM2-ES. We used the representative concentration pathways scenarios (RCP) 4.5 and 8.5 associated with an average and high concentration of greenhouse gases (IPCC, 2014). Regarding the global average temperature until the end of the 21st century, scenario 4.5 predicts an average increase in temperature of 1.1–2.6°C, while scenario 8.5 predicts an average increase of 2.6–4.8°C. Future climatic data was obtained from the Global Climate Model (Navarro-Racines et al., 2020).
To avoid problems associated with predictor multicollinearity, we used a Principal Component Analysis (PCA) to synthesize the variation of environmental predictors into orthogonal principal components (Pearson, 1901). Before running the PCA, we standardized the environmental predictors using z-scores. Then, we used the first nine principal components (Tables S4, S5), which explained ca. 95% of the original variation. Using the climatic data for the future scenarios for each AOGCM, together with the soil variables (kept as constant from the current projection), we projected the principal components for all RCPs and AOGCMs.
Land-use dataWe obtained land-use data for current (2012) and future (2050) scenarios at a 500m resolution from Soares-Filho et al., (2016). Land-use projection for 2050 was built using the OTIMIZAGRO model, a spatially-explicit model that uses climatic suitability for nine annual crops and probabilities of native vegetation loss, based on historical trends between 1994 and 2013 (MAPA, 2014). Land conversions were constrained to areas where conversion complied the environmental legislation (Soares-Filho et al., 2016). From these maps, “grassland”, “grassland in protected areas”, “savanna”, and “savanna in protected areas” were considered as native vegetation.
Modeling proceduresIn order to model species with few occurrences and overcome overfitting (i.e. having more predictors than occurrence points), we used ensembles of small models (ESM) (Breiner et al., 2015). ESM constructs a series of bivariate models and tests all the different combinations of pairs of predictors. These bivariate models are later combined in an ensemble model, avoiding overfitting without losing the ability to estimate the species niche (Breiner et al., 2015; Lomba et al., 2010). We used the Maximum Entropy (MAXENT) algorithm, which creates robust models that are transferable to other regions according to the ESM methodology and do not require excessive computational time (Breiner et al., 2018). As only presence data were available, and MAXENT needs background points, we created five sets of 10,000 random points in the accessible area for each species. Biogeographic provinces sensuLöwenberg-Neto (2014) defined the accessible areas where a species was recorded.
Models were validated by randomly dividing occurrence data into 70–30% for model training and testing, respectively; this partitioning was repeated five times. Prevalence was set to 0.5, meaning that the presences will have the same importance as the absences in model calibration. We evaluated each bivariate model with AUC and Boyce's index (Breiner et al., 2015) because they are threshold-independent and are complementary, since the first needs presence and absence data, while the second only needs presence data (Hirzel et al., 2006; details in Appendix S2). Models selected for the ensemble were projected for current and future scenarios (AOGCMs and RCPs) for 2050. For the elaboration of ensemble models for current and future scenarios, we used the Somers’ D values to weight the models in the ensemble averaging. Then we selected the bivariate models with Somers’ D >0 to compose the ensemble, because they represent models better than random (Breiner et al., 2015). We binarized the ensemble models using a threshold that included 100% of the occurrence for species with <50 points, also known as Lowest Presence Threshold (Pearson et al., 2007), and 80% for the species with >50 points to avoid overestimating species range.
To reduce overprediction in distribution ranges, we constrained current species distribution by a minimum convex polygon considering a 50km buffer around occurrence area (Mendes et al., 2020). Subsequently, we discarded areas without native vegetation from species distribution. Future species distributions were assessed using two dispersal scenarios: “no-dispersal” and “dispersal”. The no-dispersal scenario assumed that future species distribution would be in areas where present and future conditions overlap with native vegetation within the current species distribution. In the dispersal scenario, a dispersal rate of 1kmyr−1 was assumed until 2050 from the border of current distribution, based on African palm species (Blach-Overgaard et al., 2015). Thus, we made a 38km buffer around the current distribution and only considered future suitable areas in native vegetation. We calculated the percentage of loss or gain of distribution for current and future conditions given the two dispersal scenarios. For this, we divided the area of each RCP of the future dispersal and no-dispersal scenarios over the current total area. We calculated rates of species distribution change as the ratio between future and current species distribution.
We evaluated how species range changes varied according to different RCPs, dispersal scenarios, and species groups (i.e. typical and associated palm species) using generalized additive models for location, scale, and shape (GAMLSS; Rigby et al., 2019; details in Appendix S3). We developed the distribution models using the package “ecospat” (Di Cola et al., 2017) and performed GAMLSS using the package “gamlss” (Rigby and Stasinopoulos, 2005) in software R v.3.6.1 (R Core Team, 2019; further information in Appendix S4).
Protected areasIn order to assess current and future species protection degree, i.e. the degree of representativeness of a species range within PAs (Velazco et al., 2019), we calculated the proportion of species range that was within PAs. We considered two broad categories: strictly protected areas (SPs) and sustainable use areas (SUs). PA data were obtained from CNUC (2019).
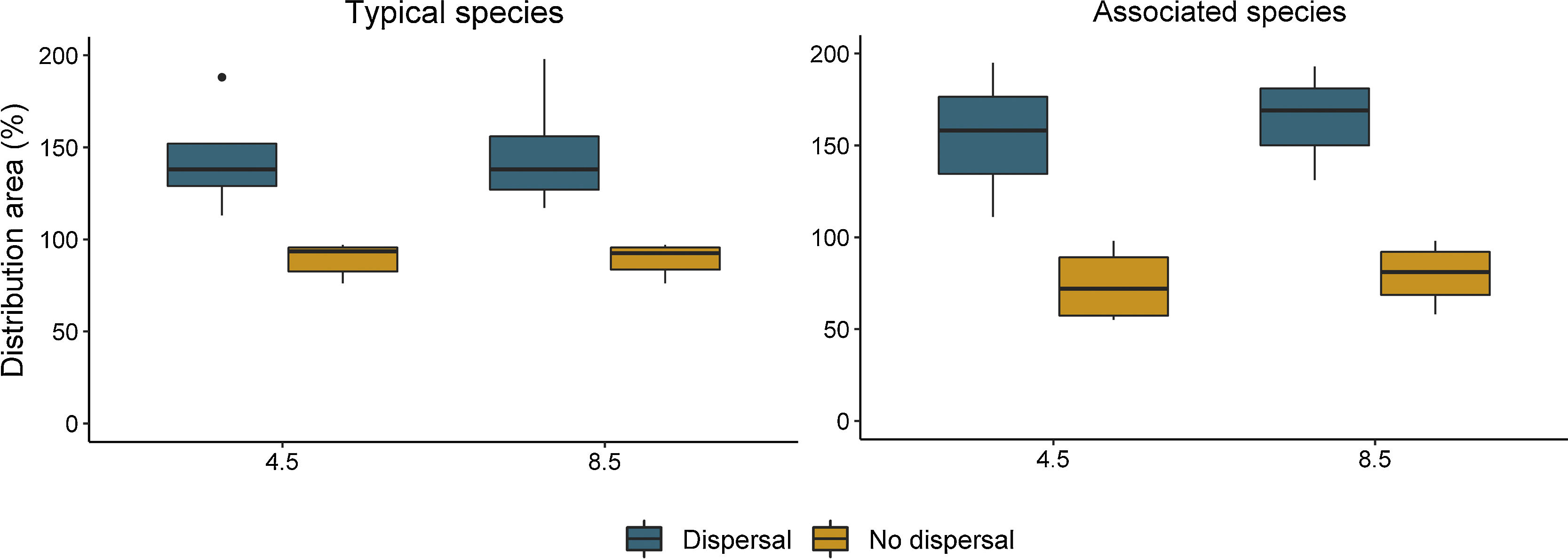
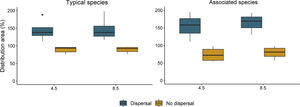
ResultsOverall, models presented a good performance, with average AUC and Boyce's index of 0.87 and 0.70, respectively (Table S6). The change in distribution area varied between dispersal scenarios and species groups (Table S7). Our future projection shows that all palm species would lose distribution area under the no-dispersal scenario and that such reduction would be greater for the associated species (Fig. 2, Figs. S3 and S4). Considering the dispersal scenario, both typical and associated species could increase their area (Fig. 2, Figs. S3 and S4).
Percentage of remaining distribution area for the typical and associated palm species of South Brazilian grasslands, based on their current distribution (100%), considering environment and land-use changes under different emission scenarios (RCPs 4.5 and 8.5) and dispersal scenarios for 2050. The outlier within the group of typical species corresponds to Butia lallemantii because the gain in area for this species is much higher than the average gain for the other species.
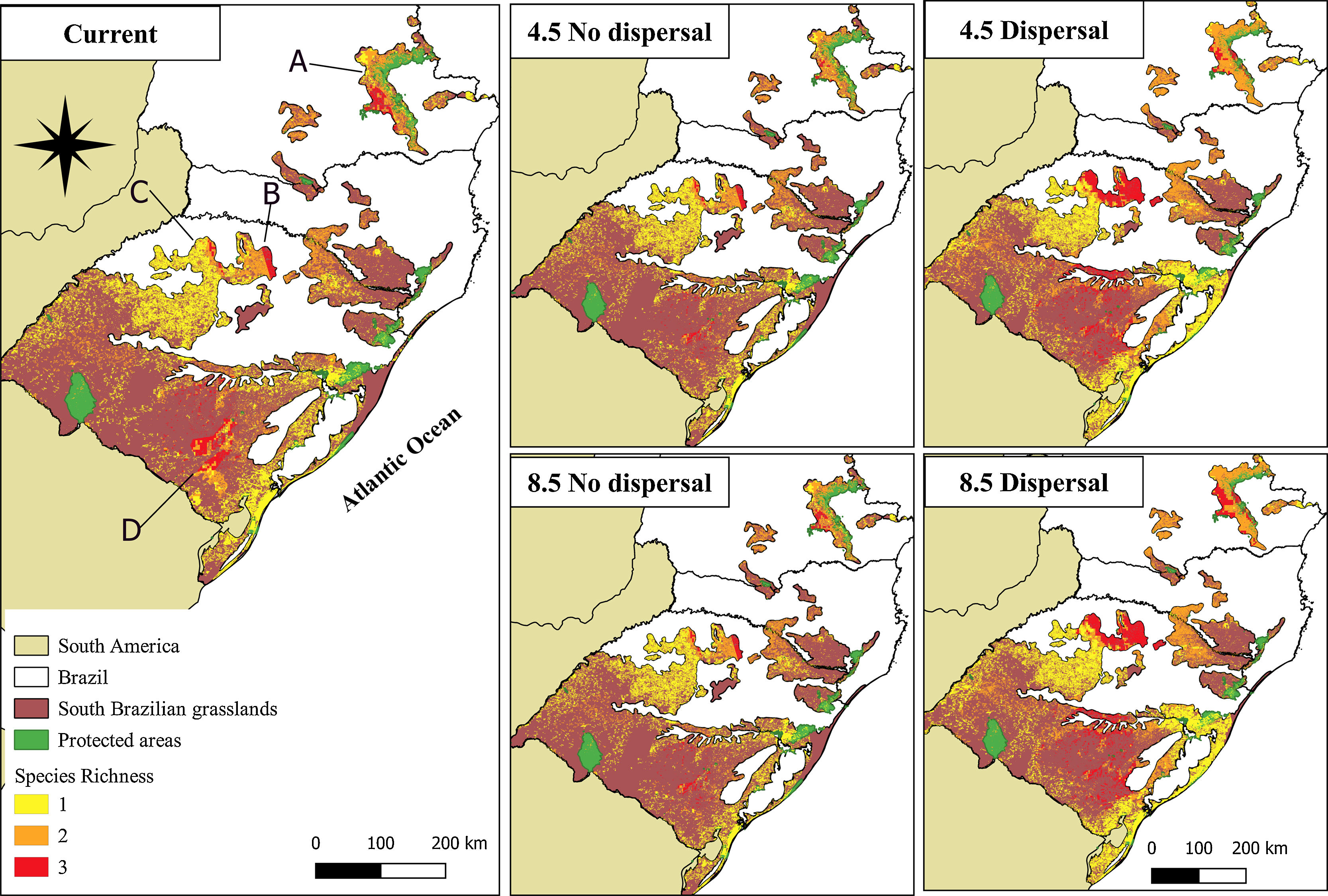
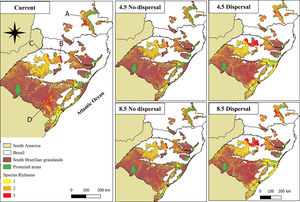
Under the current environment and land-use conditions, the highest values for richness of palm species were found in the Campos Gerais, Planalto Médio, Campos das Missões, and Serra do Sudeste (for more details about regions of South Brazilian grasslands, see Appendix S5). We predicted a reduction of species richness in areas that currently have the highest species richness in the future no-dispersal scenario. On the other hand, in the dispersal scenario, the species richness would increase in these same areas (Fig. 3, Fig. S5).
Our results indicated that the current protection degree is low, and most of the species have less than 2% of their distribution protected within strictly protected areas (SPs) and sustainable use areas (SUs), except for B. pubispatha, A. campestris and B. microspadix, which currently have, respectively, 100%, 25% and 22% of their distribution within SUs (Table 1). The results also shown that two species are gap species, i.e. they are not represented in PAs: B. witeckiiand B. paraguayensis. Considering the future dispersal scenarios, a slight increase in the protection degree of B. microspadix, B. odorata and T. acanthocoma within SUs is expected. However, the protection degree is expected to be constant in the future under both dispersal and no-dispersal scenarios for the other species (Table 1).
Protection degree of palm species distribution within strictly protected areas (SP) and sustainable use protected areas (SU) in South Brazilian grasslands (in km2 and %) under current and future scenarios of emission (RCPs 4.5 and 8.5) and different dispersal scenarios.
| Species | Current | 4.5 Dispersal | 4.5 No dispersal | 8.5 Dispersal | 8.5 No dispersal | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Total area | in SP | in SU | Total area | in SP | in SU | Total area | in SP | in SU | Total area | in SP | in SU | Total area | in SP | in SU | |
| A. campestris | 5414 | 9.25 (<1%) | 1355.75 (25%) | 10532.75 | 19.75 (<1%) | 2623 (25%) | 5287 | 9.25 (<1%) | 1314 (25%) | 10472.25 | 18.75 (<1%) | 2591.75 (25%) | 5288.25 | 9.25 (<1%) | 1315.25 (25%) |
| B. eriospatha | 10208.25 | 50.5 (<1%) | 18 (<1%) | 19218.25 | 63 (<1%) | 94.75 (<1%) | 9902.75 | 50.5 (1%) | 17.25 (<1%) | 20162.5 | 64.75 (<1%) | 121 (1%) | 9950 | 50.5 (1%) | 17.25 (<1%) |
| B. exilata | 4743.25 | 4.5 (<1%) | 0 | 6095.5 | 5 (<1%) | 0 | 4468.25 | 4.5 (<1%) | 0 | 6044.25 | 5 (<1%) | 0 | 4450.5 | 4.5 (1%) | 0 |
| B. lallemantii | 3229.75 | 0 | 73.25 (2%) | 8137.5 | 0 | 101 (1%) | 2992 | 0 | 59.75 (2%) | 6638.25 | 0 | 101 (2%) | 2940.75 | 0 | 59.75 (2%) |
| B. microspadix | 6801 | 58.75 (1%) | 1477.5 (22%) | 10345 | 77.5 (1%) | 2572.5 (25%) | 6510.75 | 58.75 (1%) | 1432 (22%) | 10603.5 | 77.5 (1%) | 2572.5 (24%) | 6561.75 | 58.75 (1%) | 1432 (22%) |
| B. odorata | 32349 | 558.5 (2%) | 294.5 (1%) | 44580.75 | 777.75 (2%) | 800 (2%) | 24514.5 | 530 (2%) | 254.5 (1%) | 44558 | 777.75 (2%) | 800 (2%) | 24526.25 | 530 (2%) | 254.5 (1%) |
| B. paraguayensisa | 81 | 0 | 0 | 89.75 | 0 | 0 | 44.75 | 0 | 0 | 106.25 | 0 | 0 | 58.5 | 0 | 0 |
| B. pubispathab | 1 | ||||||||||||||
| B. yatay | 33925.5 | 9.5 (<1%) | 78.25 (<1%) | 38432.25 | 15.5 (<1%) | 78.25 (<1%) | 26632.75 | 9 (<1%) | 47 (<1%) | 39566 | 15.5 (<1%) | 91.75 (<1%) | 27313.25 | 9 (<1%) | 55.5 (<1%) |
| B. witeckiia | |||||||||||||||
| T. acanthocoma | 11846 | 159 (1%) | 99.25 (1%) | 18764.75 | 179.25 (1%) | 293 (2%) | 10232.25 | 153.5 (2%) | 88.25 (1%) | 20060 | 182.5 (1%) | 540.5 (3%) | 10665 | 155 (1%) | 88.25 (1%) |
| T. brasiliensis | 6993.75 | 4.25 (<1%) | 1.75 (<1%) | 18571 | 58.5 (<1%) | 0 | 4070 | 3.75 (<1%) | 0 | 18361.5 | 58.5 (<1%) | 0 | 4070 | 3.75 (<1%) | 0 |
Gap species, i.e. those whose distribution is not found within any protected area (Rodrigues et al., 2004).
The synergistic effects of climate and land-use changes under the no-dispersal scenario might reduce the future geographical distribution of palms in southern Brazilian grassland. It is important to clarify that the decrease in the distribution area of associated palm species within the study area does not necessarily mean a decrease in the total area of these species, because they also occur in other areas such as the Cerrado, mixed Araucaria forests, rocky outcrops of the Pampa biome and riparian forest edges (Cano et al., 2013; Morais, 1996; Flora do Brasil 2020 em construção). On the other hand, the scenarios with dispersal show that it is possible species distributions will increase. A similar pattern of decrease in area when constraining the dispersal and increase under a dispersal scenario was also observed for palm species in Africa (Blach-Overgaard et al., 2015), monkeys in the Amazon (Sales et al., 2019), and Neotropical palms (Velazco et al., 2020). Based on our results, we confirm our first and second hypothesis. It is important to clarify the meaning of the no-dispersal and dispersal scenarios used here: the no-dispersal scenario seems feasible for all studied species, considering the past and current trends of habitat loss they are experiencing (Table S1). The no-dispersal scenario seems especially feasible for Butia exilata, which, in landscapes under severe land use change, is increasingly confined to roadsides, i.e. to the very last remnants of natural grasslands (authors’ observ.). Regarding the scenario with dispersal, we used the only information currently available (to our knowledge) on dispersal rates of palms, which was done for African species (Blach-Overgaard et al., 2015), thus our scenario with a dispersal of 1kmyr−1 might be optimistic. However, geometrically speaking, the more a species is able to disperse, the more its range will increase. Then, if we generated a new scenario of intermediate dispersal (e.g. 500myr−1), the increase in the range would lay somewhere in between the no-dispersal and the dispersal scenarios. The most important point to make about the implications of these scenarios is that factors such as habitat loss, defaunation and overexploitation contribute to a result closer to the no-dispersal scenario, while the creation of PAs and improvement of grassland management and conservation contribute to the dispersal scenario. Recent findings indicate that, among rare species confined to particular microhabitats, dispersal distance can play a fundamental role in species persistence (Di Musciano et al., 2020), so it will be important to obtain real estimates of species dispersal abilities in future studies.
The gain in distribution area under the dispersal scenario is overly optimistic because it would mean the expansion of species distribution to new territories. Actually, this is unlikely to happen, because the ability of palm species to disperse and occupy new areas depend on other factors, such as (i) the capacity of pollination and seed dispersal agents, such as birds and mammals (Nascimento et al., 2020), to positively respond to climate change; (ii) the negative biotic interactions of the resident community with the newcomers (Corlett, 2016), such as competitive exclusion, predation, and parasitism; and (iii) on land management (e.g. grazing pressure) (Sosinski et al., 2019). There is evidence that, as the defaunation increases in fragmented landscapes, the recruitment and regeneration of palm populations decrease (Galetti et al., 2006), and that long-term homogenization of genetic pools of palm populations may occur (Carvalho et al., 2016). Moreover, the dispersal scenario assumes that future landscape will follow the projections made by Soares-Filho et al. (2016), i.e. land occupation complying with the environmental law – an unlikely situation considering Brazil's economic expansion policies (Loyola, 2014) and current setbacks in national environmental legislation enforcement (Abessa et al., 2019).
Our results highlight the low protection degree of most palm species regarding their distribution in the current PA network, thereby partly corroborating our third hypothesis, and reflecting the negligence in protecting the South Brazilian grasslands (Overbeck et al., 2015b, 2007). A similar result was found for Neotropical palms, under different dispersal scenarios, in which the existing PA network had a poor performance in protecting the phylogenetic diversity of palms under current and future climates (Velazco et al., 2020). A clear implication of the low protection degree of studied palm species can be seen for B. witeckii and B. paraguayensis, considered as gap species because they are not represented in any PAs. It is also noteworthy that the occurrences of palm species also depend on the management grasslands are subject to. Since most of the protection of species is on PAs of SU, palm conservation should be carefully planned to avoid lack of regeneration under high grazing pressures, as these PAs allow sustainable management and use of natural resources. For example, appropriate livestock management rotation in butiazal1 ecosystems enabled the establishment and growth of seedlings of B. odorata (Sosinski et al., 2019). Such management consisted in selecting butiazal areas to exclude from grazing during the winter and allow low density herds to graze beneath the palm grove during the other seasons (Sosinski et al., 2019; Rivas, 2005). In an opposite, drastic situation, the only known population of B. pubispatha is 100% encompassed by the Environmental Protected Area of Escarpa Devoniana (a SU). However, this unique population is mostly covered by a recent plantation of Pinus sp. (authors’ observ.), which can lead the species to extinction if no urgent actions are taken, e.g., the formation of seed banks, live collections, and assisted migration. Furthermore, although B. microspadix has 22% of its distribution within SUs, care must be taken because this species is endemic to the Campos Gerais region (Fig. S5), where it is considered vulnerable because of increasing habitat loss.
Moreover, the lack of management in most of the strictly protected areas in South Brazilian grasslands (Overbeck et al., 2007) also seems to threaten palm species typical of grassland environments. The population of B. exilata, which is found in the State Park of Papagaio-Charão (a SP) in the Planalto Médio region (Fig. S5), seems to be in decline due to forest expansion over grassland habitats of this species (authors’ observ.). Forest expansion over grasslands has been observed throughout southern Brazil (Behling, 2002; Oliveira and Pillar, 2004; Quadros and Pillar, 2001), therefore grazing and fire management can be a double-edged sword for Butia palm groves because the prevention of forest expansion is balanced by the possible negative effect of higher grazing pressure or intensive fire use on palm recruitment (Sosinski et al., 2019). Further research is needed to understand the ecological relationship of South Brazilian grasslands with fire and grazing (Overbeck et al., 2015a) and implement adequate management that promotes palm species persistence.
Studying the combined effect of climate and land-use changes in the distribution of palm species under different dispersal scenarios enabled exploring a wide range of future possibilities for these plants. We observed that if palm species are able to disperse, the synergistic effects of climate and land-use changes may not be bad for them. However, as we did not consider other factors in the dispersal scenario, such as local adaptation of palm species to non-climatic conditions (e.g. like relief, slope, soil chemical characteristics), and the responses of pollinators and seed dispersers to global changes, it is unlikely that palm species will disperse in the assumed dispersal rates. For this reason, the no-dispersal scenario seems to be more plausible, which points to the necessity of the implementation of urgent conservation actions, as these species are mostly endangered due to habitat loss. Most of the studied species are restricted to South Brazilian grasslands, where they are also economically important as they are used as raw material in handicrafts, food production, and as ornamental plants (Büttow et al., 2009; De Lourdes et al., 2007; Sosinski et al., 2019), which could increase the pressure on their populations, or alternately stimulate their protection through public awareness. Besides the impacts on the palm populations per se and on the associated fauna, species loss might also impact the regional economy. Our analyses revealed that PAs provide low protection to palm species and highlighted which of them are gap species, which can inform the formulation of conservation policies and support the creation of new PAs in the study region. Further research is needed to expand the knowledge on the distribution and biology of palm species, especially B. pubispatha,B. witeckii and B. exilata, which have only a few known populations and need more information to properly plan for their conservation.
Finally, we suggest some actions that can be taken to protect palm species of the South Brazilian grasslands. In-situ actions that could be taken involve the development of policies aiming to reduce the conversion of the South Brazilian grasslands, the expansion of the current PA network, adequate grazing and fire management allowing the recruitment of palm species, restoration of palm groves through the introduction of new seedlings (Sosinski et al., 2019), and the incentive of sustainable use of fruits and leaves. Regarding adequate management, we suggest initiatives to train and support landowners to maintain and properly manage palm groves in their legal reserves, with which private lands must comply (Brancalion et al., 2016). Ex-situ actions important to consider include the formation of seed banks, cryopreservation, live collections, and assisted migration (Corlett, 2016), especially for rare and geographically restricted species, which might increase the availability of seedlings available to restore palm groves. Putting these actions in practice is necessary to ensure the persistence of these palm species and their ecosystem in the future.
Concluding remarksThis study presented the first evaluation of the impact of climate and land-use changes on the geographical distribution of palm species in the South Brazilian grasslands and assessed the protection degree of these species in the current network of PAs considering present and future species distributions. We show that the scenario where species will be unable to migrate to new areas (no-dispersal) is unfortunately the more plausible scenario under increasing rates of land use changes. Furthermore, new protected areas should be created to increase protection to palm species in the future. Our findings reflect the state of conservation of South Brazilian grasslands and invite stakeholders to develop actions towards the protection of palm species and their ecosystems.
FundingThis work was supported by the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior – Brasil (CAPES) – Finance Code 001, under a master's scholarship to LFCT and EMG, and a post-doctoral fellowship to FTB (PNPD/CAPES, process number 88882.306081/2018-1). Financial support was also obtained from a postdoctoral fellowship by Consejo Nacional de Investigaciones Científicas y Técnicas (CONICET/Argentina) to SJEV, a doctoral scholarship by Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq/Brazil) to PMH (#141137/2020-7), and a grant by the Fundação Grupo Boticário de Proteção à Natureza (project #1124_20181).
We thank Priscila Lemes, Cíntia Freitas, Marília Borgo, two anonymous reviewers, and Gerhard Overbeck for important comments on previous versions of this paper.